Protocol - Arrhythmia (Atrial and Ventricular)
Description
Measure to assess presence of arrhythmias through collection of personal history, treatments and procedures, medication usage, and administration of an electrocardiograph (ECG).
Specific Instructions
Ask question 1, and if "Yes," continue through protocol. Completing an electrocardiograph will provide additional specificity for the measure. If question 1 is "No," the Working Group recommends not proceeding with the protocol.
Availability
This protocol is freely available; permission not required for use.
Protocol
1. Have you been told you have/had a heart rhythm problem called atrial fibrillation?
[ ] 0 No
[ ] 1 Yes
[ ] 2 Maybe
[ ] 9 Unknown
If yes:
2. Date of first episode __ / __ / ____
[ ] 99/99/9999 Unknown
code year as 4 digits, example: Year 1999=1999
3. ER/hospitalized or saw M.D.?
[ ] 0 No
[ ] 1 Hosp/ER
[ ] 2 Saw M.D.
[ ] 9 Unknown
Hospitalized at:__________________________________
M.D. seen: _____________________________________
4. Permanent pacemaker insertion?
[ ] 1 Yes
[ ] 0 No
[ ] 2 Maybe
[ ] 9 Dont Know
If Yes:
Year done: __ __ __ __
[ ] 9999 Unknown
5. Are you taking any of the cardiovascular medications below?
a. Anticoagulants (Coumadin, Warfarin, etc.)
[ ] 0 No
[ ] 1 Yes, now
[ ] 2 Yes, not now
[ ] 3 Maybe
[ ] 9 Unknown
b. Antiarrhythmics (Quinidine, Procainamide, Norpace, Disopyramide, etc.)
[ ] 0 No
[ ] 1 Yes, now
[ ] 2 Yes, not now
[ ] 3 Maybe
[ ] 9 Unknown
6.0 JACKSON HEART STUDY CLINIC ELECTROCARDIOGRAPH (ECG) PROCEDURES
6.1 Introduction
At the baseline visit, a standard supine 12-lead resting ECG is recorded after a 12-hour fast and at least one hour after smoking or ingestion of caffeine, followed by a light snack.
6.2 Procedures for Recording ECGs
A supine twelve lead digitized ECG will be the standard for the Jackson Heart Study, and the MAC PC Personal Cardiographic Computer by Marquette Electronics, Inc., is recommended for the study. The MAC PC is a widely used ECG machine that is approved by the Underwriters Laboratory and has been used extensively in other epidemiologic studies. The MAC is the most advanced cardiograph available today. For the JHS, the function of the MAC PC will be used to obtain and store ECG data prior to transmission to the ECGRC. Complete instructions for operating the MAC PC are provided by the company and are easy to understand.
The standard configuration for the MAC PC is shown in Appendix 2. A 12-lead resting ECG tracing is obtained consisting of 10 seconds of each of the leads simultaneously (I, II, III, aVR, aVL, aVF, Vl-V6).
6.3 Electrode Position Measuring and Marking
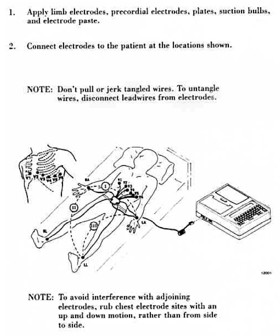
A standard procedure will be followed for location of chest electrode sites and limb leads.
Measurements taken for chest electrodes will be used for quality control. This protocol will mimic that used in the ARIC Study. The electrodes are attached to the lead wires of the acquisition module in the correct order by using the lead wire labels. Simultaneously with the digital recording, a paper record is produced that can be inspected for data quality, lead reversal and alert ECG statements.
Because it is essential for the study to be able to compare baseline ECG data with subsequent records, a uniform procedure for electrode placement and skin preparation is required. The method and procedure for standardizing electrode locations are outlined below. The participant, chest bared, is instructed to lie on the recording bed with arms relaxed at the sides. The individual is asked to avoid movements, which may cause errors in marking the electrode locations, but encouraged to converse with the technician. Prior experience with electrocardiograms is discussed, as is the purpose of the ECG recording. The participant should be told this is a research ECG to be used for statistical analysis later in the study. However, the clinic physician can also use it for general diagnostic purposes, and a copy can be sent to the individuals private physician if desired. For best electrode/skin interface, place the electrodes on the skin at least 2-3 minutes before taking the ECG. Participant information can be entered on the MAC PC during this time.
A good felt tip pen is used to mark the six chest electrode positions. Wipe the general area of the following 10 electrode sites with a sterile alcohol prep to remove skin oil and perspiration. It is extremely important that care be taken to locate these positions accurately. Therefore, the procedure given below must be meticulously followed. Electrode positions in women with large, pendulous breasts must be determined in relation to the anatomic points described below, as for all participants. The electrodes must then be placed on top of the breast (in the correct position). Figure 1 schematically displays the standard electrode locations.
6.3.1 Limb Leads
Locate electrode LL on the left ankle (inside).
Locate electrode RL on the right ankle (inside).
Locate electrode LA on the left wrist (inside).
Locate electrode RA on the right wrist (inside).
6.3.2 Electrode V2
Locate the sternal angle and second left rib between the index and middle fingers of your right hand. Count down to the fourth rib and identify the fourth intercostal space below it. Locate V2 in the fourth intercostal space immediately to the left of the sternal border.
6.3.3 Electrode V1
Locate electrode V1 in the fourth intercostal space at the right sternal border. This should be at the same level as V2 and immediately to the right of the sternum.
6.3.4 Anterior 5th Interspace Marker (E Point)
Identify the fifth rib and fifth intercostal space below V2 by counting down ribs as described for V2. Follow this space horizontally to the midsternal line and mark this point. This is the "E" point.
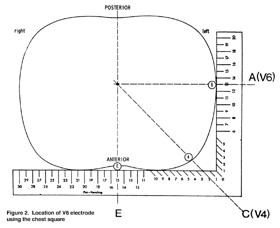
6.3.5 Electrode V6
With the chest square held lightly against the body (see Figure 2) locate the V6 electrode at the same level as the E point in the midaxillary line (straight down from the center of the armpit). If breast tissue is over the V6 area, mark the V6 location on the breast. Do not attempt to move the breast in order to mark V6 on the chest wall.
6.3.6 Chest Size Measurements
Place the Chest Square firmly on the lower sternum at location E and at location V6. Verify that the arms of the square are exactly horizontal and vertical in the horizontal plane of the thorax. Move the square so that the vertical arm at V6 is firmly against the ribcage. Use the appropriate length chest square so the horizontal arm rests firmly on the lower sternum.
Now read the distance 0-E and the distance 0-V6 to the nearest 0.5 cm. Write them down on scratch paper.
Record the 0-V6 measurement under height and the O-E measurement under weight. Measure to the nearest 0.5 cm. and round up. e.g., 11.25 cm. would be 11.5 cm. 11.75 cm. would be 12.0 cm.
Enter 3 digits into the MAC PC but do not enter decimal points.
e.g., 11.5 cm enter as 115 11 cm. enter as 110
Use leading zeros.
e.g., 9.5 cm. enter as 095
6.3.7 Electrode V4
Electrode V4 is located using the E-V6 Halfpoint Method 3. Using a flexible tape measure, measure the distance between the E point and the V6 marking. The tape should be resting lightly on the skin, not pressing into the flesh. The E and V6 marks should clearly be seen above the tape. Without moving the tape, mark the location of electrode V4 midway between E and V6.
6.3.8 Electrode V3
Using a flexible tape measure, mark the location of electrode V3 midway between the locations of V2 and V4.
6.3.9 Electrode V5
Using a flexible tape measure, mark the location of electrode V5 midway between the locations of V4 and V6.
6.4 Skin Preparation
Prepare the skin for applying electrodes by wiping with alcohol, then briskly with a gauze pad. If technical problems are observed due to poor electrode contact, it is necessary to do further preparation as described below:
1. With the participants consent, remove any excess hair from each electrode site on the chest and legs using an electric or disposable shaver.
2. At each electrode location in turn, the outer horny layer of the epidermis is removed by gentle dermal abrasion with a piece of 6-O (220) sandpaper. Only three passes (in the form of an asterisk) at each site using light pressure are required.
If the skin preparation has removed the felt pen marking at any of the electrode sites, these are accurately reestablished by carefully repeating the procedure described in Electrode Position Measuring and Marking. It is important that the electrode sites be marked using the exact technique described.
6.5 Application of Electrodes
Disposable electrodes are used in the JHS. Adaptors are used with the leadwires to connect the "banana" plug from the MAC PC leadwire to the disposable electrode via a clip.
When placing each electrode, massage it with a small up and down circular motion to maximize the pre-gel contact with the skin but avoid overlap of gel from one electrode to the next.
Center the four limb electrodes on the inside of the wrist or ankle with the tab for the clip pointing toward the head. Center the six chest electrodes on the chest markings with the tabs pointing down. Do not let the electrodes overlap or touch each other if possible.
Clip the appropriate leadwire to each electrode. Do not pull or jerk tangled wires. To untangle wires, disconnect lead wires from electrodes.
6.6 Recording the 12-lead ECG
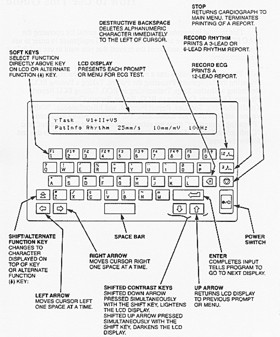
After placing the electrodes on the skin, enter the participant information into the MAC PC (Figure 3) according to Appendix 2. Electrodes must be on the skin for at least 2-3 minutes before taking the ECG. Make a final check of the electrodes and lead wires. Ask the participant to relax and keep still, then press the RECORD key.
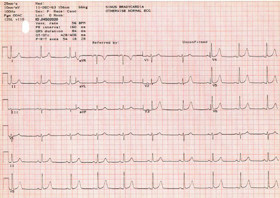
The machine will display "Acquiring Data" and the left side of the display will show a count. If there are technical problems the display will show which lead is involved and will keep counting until it gets 10 seconds of good data. Check electrode contacts and leadwires, then check the display again. If the display counts past 45, electrodes likely will need to be re-positioned. Push the STOP key and remove the electrodes on limbs first. Prepare the electrode sites as discussed in Skin Preparation and follow the above protocol for exact relocation of electrodes. Press RECORD ECG. The machine will tell you to "enter a new patient or press RECORD." Press RECORD ECG a second time to start the ECG. The machine will automatically print the ECG after it has acquired 10 seconds of good data (Figure 4).
Tear the ECG off the machine and file it in your records. Make a copy by pressing "up" arrows with Fl. Press Fl under "Storage", again press Fl under "Plot". Choose the desired tracing. Press F5 - "Enter" - then "Print". A copy can be printed from the machines memory any time before deletion of the ECG.
Note: Although the Jackson Heart Study uses the Minnesota Central Electrocardiographic Laboratory (ECGRC) to read and interpret ECG results, the Working Group recommends that the results can either be read manually or by computer.
Personnel and Training Required
To complete the Framingham Heart Study (FHS) questions, an interviewer who is trained to conduct personal interviews with individuals from the general population is required. The interviewer must be trained and found to be competent (i.e., tested by an expert) at the completion of personal interviews.* The interviewer should be trained to prompt respondents further if a "dont know" response is provided.
* There are multiple modes to administer this question (e.g., paper-and-pencil and computer-assisted interviews).
To complete the electrocardiograph (ECG), a certified and trained technician is required. This person will know how to reliably position electrodes, how to recognize alert conditions, and understand the quality requirements. Results from the ECG can be obtained by having a digital ECG read by computer at the Minnesota Central Electrocardiographic Laboratory (ECGRC) or by using commercially available software programs to provide a real time interpretation.
Equipment Needs
Supine 12-lead digitized electrocardiograph (ECG) machine, MAC PC Personal Cardiographic Computer by Marquette Electronics, Inc.® , felt-tip pen, sterile alcohol prep pads or alcohol and gauze pads, chest square, flexible tape measure, electric or disposable shaver
Requirements
| Requirement Category | Required |
|---|---|
| Major equipment | Yes |
| Specialized training | Yes |
| Specialized requirements for biospecimen collection | No |
| Average time of greater than 15 minutes in an unaffected individual | No |
Mode of Administration
Interviewer-administered question, Clinical measurement
Lifestage
Adult
Participants
Framingham Heart Study (FHS): ages 19–79 years
Jackson Heart Study (JHS): ages 35–84 years
Selection Rationale
These combined protocols collect information about a persons heart rhythm, including personal history, treatments and procedures, medication usage, and a clinical measurement. This combination is sufficient to assess history of or current presence of arrhythmias.
Language
Chinese, English, Other languages available at source
Standards
| Standard | Name | ID | Source |
|---|---|---|---|
| Logical Observation Identifiers Names and Codes (LOINC) | PhenX - arrhythmia protocol | 62400-7 | LOINC |
| Human Phenotype Ontology | Arrhythmia | HP:0011675 | HPO |
| caDSR Form | PhenX PX041101 - Arrhythmia Atrial And Ventricular | 5837955 | caDSR Form |
Derived Variables
None
Process and Review
Not applicable.
Protocol Name from Source
Framingham Heart Study (FHS), Exams 6 & 29 & Jackson Heart Study (JHS)
Source
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute and Boston University. Framingham Heart Study (FHS). Exam 29. Medical History. Pages 36, 40. No question numbers provided (source for questions 1–4).
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute and Boston University. Framingham Heart Study (FHS). Exam 6. Medical History—Cardiovascular Medications. Page 19. No question numbers provided (source for questions 5a and 5b).
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute, National Center on Minority Health and Health Disparities, and the National Institute of Biomedical Imaging and Bioengineering. Jackson Heart Study (JHS). Manual 5. Electrocardiography. Visit 1. Version 1.0. February 15, 2001. No question number provided (source for question 6).
General References
None
Protocol ID
41101
Variables
Export Variables| Variable Name | Variable ID | Variable Description | dbGaP Mapping | |
|---|---|---|---|---|
| PX041101_Atrial_Fibrillation_ER_Hospitalized_MD | ||||
| PX041101030000 | Have you been told you have/had a heart more | Variable Mapping | ||
| PX041101_Atrial_Fibrillation_Hospitalized_At | ||||
| PX041101030100 | Hospitalized at | N/A | ||
| PX041101_Atrial_Fibrillation_MD_Seen | ||||
| PX041101030200 | M.D. seen | N/A | ||
| PX041101_Cardiovascular_Medication_Antiarrhythmic | ||||
| PX041101050200 | Are you taking any of the cardiovascular more | Variable Mapping | ||
| PX041101_Cardiovascular_Medication_Anticoagulant | ||||
| PX041101050100 | Are you taking any of the cardiovascular more | Variable Mapping | ||
| PX041101_ECG_Image_ID | ||||
| PX041101060100 | Electrocardiogram Trace/Image ID. | N/A | ||
| PX041101_ECG_Other | ||||
| PX041101060200 | Other, specify | N/A | ||
| PX041101_Ever_Have_Atrial_Fibrillation | ||||
| PX041101010000 | Have you been told you have/had a heart more | Variable Mapping | ||
| PX041101_First_Episode_Date | ||||
| PX041101020000 | Date of first episode (mm/dd/yyyy) | Variable Mapping | ||
| PX041101_First_Episode_Date_Coded | ||||
| PX041101020100 | Date of first episode (mm/dd/yyyy) | N/A | ||
| PX041101_Pacemaker | ||||
| PX041101040100 | Permanent pacemaker insertion? | Variable Mapping | ||
| PX041101_Pacemaker_Year | ||||
| PX041101040200 | Year inserted permanent pacemaker. | N/A | ||
| PX041101_Pacemaker_Year_Coded | ||||
| PX041101040201 | Year inserted permanent pacemaker. | N/A | ||