Protocol - Child Head Circumference
- Birth Weight - Birth Weight Abstracted from Medical Records
- Birth Weight - Measured Weight at Birth
- Birth Weight - Proxy Reported Birth Weight
- Growth Charts
- Current Age
- Ethnicity and Race
- Height - Knee Height
- Height - Recumbent Length
- Height - Self-Reported Height
- Height - Standing Height
- Weight - Measured Weight
- Weight - Self-Reported Weight
Description
Measurement for children from birth to 36 months (Centers for Disease Control and Prevention growth charts) or 60 months (World Health Organization growth charts) of age. The parent or a health professional may need to hold the child while the examiner uses a tape measure to measure the child’s head.
Specific Instructions
Several overarching, critical issues for high-quality data collection of anthropometric measures that optimize the data in gene-environment etiologic research include (1) the need for training (and retraining) of study staff in anthropometric data collection; (2) duplicate collection of measurements, especially under field conditions; (3) use of more than one person for proper collection of measurements where required; (4) accurate recording of the protocols and the measurement units of data collection; and (5) use of required and properly calibrated equipment. The PhenX Anthropometrics Working Group recommends that this protocol also be used on participants from birth to 36 months of age (see Centers for Disease Control and Prevention growth charts).
The Expert Review Panel notes that this protocol could be used for children up to 60 months of age if there are environmental concerns (e.g., malnutrition; see World Health Organization growth charts).
Notes from the Expert Review Panel: The notion of recommending replicate measurements comes from the reduction in random errors of measurement and accompanying improved measurement reliability when the mean of multiple measurements is used rather than a single measurement. This improvement in measurement reliability, however, depends on the reliability of a single measurement in the hands of the data collectors in a particular study (Himes, 1989). For example, if a measure such as recumbent length in a given study has a measurement reliability of 0.95 (expressed as an intraclass correlation coefficient), taking a second measurement and using the mean of the two measurements in analyses will improve the reliability to only 0.97, yielding only a 2% reduction in error variance for the additional effort. If, in the same study, the reliability of a single triceps skinfold measurement was 0.85, using the mean, including a replicate measurement, would raise the reliability to 0.92 and yield a 7% reduction in error variance, more than a three-fold improvement compared with recumbent length. The intraclass correlation coefficient is specifically recommended here for assessing reliability because it takes account of random and systematic errors of measurement, whereas the interclass correlation (e.g., Pearson correlation) takes account of only random errors of measurement.
Because the benefits of taking replicate measurements are so closely linked with the existing measurement reliability, it is recommended that as a part of the training of those who will be collecting anthropometry data, a reliability study be conducted that will yield measurement reliability estimates for the data collectors, protocols, settings, and participants involved in that particular study (Himes, 1989). If the measurement reliability for a single measurement is greater than or equal to 0.95, the recommendation is that replicate measurements are not necessary and will yield little practical benefit. If the measurement reliability is less than 0.95, the recommendation is to include replicate measurements as prescribed.
If replicate measurements are indicated because of relatively low reliability, a second measurement should be taken, including repositioning the participant. A third measurement should be taken if the first two measurements differ by more than 0.5 cm. If it is necessary to take a third measurement, the two closest measurements are averaged. Should the third measurement fall equally between the first two measurements, all three should be averaged.
Availability
This protocol is freely available; permission not required for use.
Protocol
The following protocol is part of an examination survey. The National Health and Nutrition Examination Survey (NHANES) has collected head circumference for infants and children. The ages of the participants have varied. NHANES 1999 to 2013 collected head circumference on infants from birth to 6 months. Depending on the NHANES cycle, administrative factors may have resulted in different age groups measured. The circumference of the head is measured on children from birth through 36 months of age. Follow the steps below to obtain the head circumference measurement:
1. Position of the study participant: Instruct the parent (or guardian) to stand holding the child over the parent’s left shoulder or else sit with the child in the parent’s lap. Ask the parent to remove hair ornaments or braids that might interfere with the measurement.
2. Taking the measurement: Place the head circumference tape around the child’s head so that the tape lies: across the frontal bones of the skull; slightly above the eyebrows; perpendicular to the long axis of the face; above the ears; and over the occipital prominence at the back of the head (Exhibit 1). Move the tape up and down over the back of the head to locate the maximal circumference. Tighten the insertion tape so that it fits snugly around the head and compresses the hair and underlying soft tissues. Measure the circumference to the nearest 0.1 cm.
3. Record the result. Remove the head circumference tape.
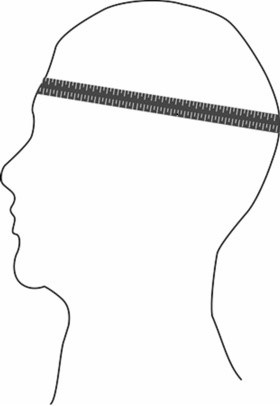
Exhibit 1. Head circumference tape position
Interpretation of Findings Current growth charts, including head circumference, can be found on the Centers for Disease Control and Prevention, National Center for Health Statistics website available at http://www.cdc.gov/growthcharts/ (accessed December 12, 2014).
Personnel and Training Required
Technicians who have experience working with young children and who should be trained in the basic techniques of anthropometric measurements
Equipment Needs
Flexible measurement tape
Requirements
| Requirement Category | Required |
|---|---|
| Major equipment | No |
| Specialized training | No |
| Specialized requirements for biospecimen collection | No |
| Average time of greater than 15 minutes in an unaffected individual | No |
Mode of Administration
Physical Measurement
Lifestage
Infant, Toddler
Participants
Infants from birth to 36 months (Centers for Disease Control and Prevention growth charts) or up to 60 months of age, based on World Health Organization standards available at [link[www.who.int/childgrowth/standards/en/|http://www.who.int/childgrowth/standards/en/]]
Selection Rationale
The child head circumference protocol from the 2007-08 National Health and Nutrition Examination Survey was selected as best practice methodology and is the most widely used protocol to assess child growth and development.
Language
Chinese, English, Spanish
Standards
| Standard | Name | ID | Source |
|---|---|---|---|
| Logical Observation Identifiers Names and Codes (LOINC) | PhenX - child head circum protocol | 62406-4 | LOINC |
| Human Phenotype Ontology | Abnormality of skull size | HP_0000240 | HPO |
| caDSR Form | PhenX PX020501 - Child Head Circumference | 5805909 | caDSR Form |
Derived Variables
None
Process and Review
The Expert Review Panel #1 reviewed the measures in the Anthropometrics, Diabetes, Physical Activity and Physical Fitness, and Nutrition and Dietary Supplements domains.
Guidance from the ERP includes:
• Added replicate measure language
• Changed unit of measurement
Back-compatible: no changes to Data Dictionary
Previous version in Toolkit archive (link)
Protocol Name from Source
National Health and Nutrition Examination Survey (NHANES), Anthropometry Procedures Manual, 2007
Source
Centers for Disease Control and Prevention, National Center for Health Statistics. (2007). National Health and Nutrition Examination Survey (NHANES) Anthropometry Procedures Manual. Hyattsville, MD: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Retrieved from http://www.cdc.gov/nchs/nhanes/nhanes2007-2008/manuals07_08.htm
Certification for the Spanish translation can be found here.
General References
None
Protocol ID
20501
Variables
Export Variables| Variable Name | Variable ID | Variable Description | dbGaP Mapping | |
|---|---|---|---|---|
| PX020501_Child_Head_Circumference_1 | ||||
| PX020501010000 | Child Head Circumference measured in more | N/A | ||
| PX020501_Child_Head_Circumference_2 | ||||
| PX020501020000 | Child Head Circumference measured in more | N/A | ||
| PX020501_Child_Head_Circumference_3 | ||||
| PX020501030000 | Child Head Circumference measured in more | N/A | ||
| PX020501_Child_Head_Circumference_Average | ||||
| PX020501040000 | Child Head Circumference measured in more | Variable Mapping | ||